Real-time redox in wetland soils

Redox potential is a key metric for verifying wetland soil conditions
The Indicator of Reduction in Soils (IRIS) is a method researchers use to check redox status on a weekly basis, but it destroys five or more plastic pipes in the process and can be prone to errors due to spatial variability in microbe populations.
A new study in Agricultural & Environmental Letters highlights an adapted IRIS methodology that automates data collection, allowing researchers to see redox status in real time.
Wetlands purify water, stabilize shorelines, serve as valuable fish and wildlife habitats, and recharge groundwater. Identifying their benefits is straightforward, but identifying their reduction-oxidation status? Well, that’s a little more difficult.
Scientists in the field and the laboratory have been using a technique called Indicator of Reduction in Soil (IRIS), which Karen Castenson and Martin Rabenhorst first published in the Soil Science Society of America Journal (SSSAJ) in 2006. The technique relies on nested polyvinyl chloride (PVC) pipes, painted with an iron oxide (ferrihydrite and goethite) or manganese paint and installed in soil. To see whether the soil is in a reducing state—and therefore, a wetland—the researcher pulls tubes from the field in sequence to see how much of the paint the soil microbes have removed. Paint removal gives scientists an idea whether the soil lacks oxygen and contains enough carbon to encourage bacterial populations to turn to the alternative electron acceptors iron and manganese.

But a new project published in Agricultural & Environmental Letters (AEL) puts a spin on the traditional IRIS methodology by using a single clear tube and a rhizosphere camera to automate the process and track redox in real time (https://doi.org/10.1002/ael2.20051). Two of the authors, Olivia LeFevre and Thorsten Knappenberger, talk through the insights, adaptations, and troubleshooting that made it possible for this proof-of-concept project to come to life.
A Senior Project
Olivia LeFevre was a first-semester senior at Auburn University when she walked into Thorsten Knappenberger’s office looking for research opportunities.
“I started thinking about grad school, and I knew I wanted to work on wetlands,” LeFevre says. “Dr. Knappenberger is a soil physics professor; he’s done lots of work on wetlands, so I got in touch.”
Knappenberger had just the project for LeFevre.
In the classic IRIS setup, a researcher can compare a tube removed from the “nest” after seven days with ones removed over the following weeks to see how microbes are behaving over time. In a true wetland, saturated soils create anaerobic conditions that encourage microbes to use iron or manganese as electron receptors.

“To classify a soil as a wetland soil, the Hydric Soil Technical Standard requires that three out of five of the tubes show evidence of reduction,” Knappenberger explains.
But the technique has a couple of drawbacks. First, it relies on multiple pipes set in the soil, which means there is spatial variability in reducing conditions between the pipes. Plus, researchers only see data for four or five time points over the whole process if they assess pipes on a weekly basis.
Instead, Knappenberger and LeFevre hatched a plan to make use of a rhizosphere camera to automate the process.
“The root camera really hadn’t worked so well for roots, but I had this idea of using it with IRIS,” Knappenberger says. “When Olivia walked through the door, it was just perfect timing.”
Instead of testing it in the field, they designed a mesocosm in the lab, setting themselves up for an hour-by-hour analysis of reduction-oxidation status in their new IRIS setup.
Automating IRIS

In 2021, Scott et al. published a study in SSSAJ that found that IRIS coatings are removed from clear tubes just as well as PVC (https://doi.org/10.1002/saj2.20171). The study spurred Knappenberger to try using a clear rhizosphere tube, cut down to a bit taller than bucket height, coated on the outside with both iron and manganese paint in four broad stripes. Along with LeFevre, he installed the camera inside it, nested the rhizosphere tube in the bucket, and filled the bucket with 8 cm of washed sand and another 20 cm of wetland soil on top of that. They sourced the soil from a surface horizon (57% sand, 27% silt, 19% clay, and 1.7% soil organic carbon). They added some peat moss for good measure, creating an additional carbon source. Finally, they saturated the soil with water, feeding it from a pipe at the bottom of the bucket. They maintained a steady water level near the top of the bucket throughout the study.
There was just one problem: they wanted to take pictures on an hourly basis, but the existing camera program that was used to take root images in the field required someone to physically click the “take photo” and “save” buttons each time.
“It was physically impossible for someone to be in the lab every time we needed a photo,” LeFevre says. “So I did some research and found this Python package that moves your mouse and clicks—you can automate pretty much anything.” LeFevre wrote a program that moved the mouse, clicking the right buttons on the screen at the right times. The program saved photos automatically to a Box folder, which Knappenberger checked daily to make sure it was still running.
“We pretty much watched the images come in, and you can see the reduction as it happens,” Knappenberger says. “The difference is obvious by eye—you can tell right where reduction stops, but the paint is brown and the soil is brown. It’s actually really tough for a computer to distinguish between them.”

Knappenberger color-corrected his photos, converting the images into different color spaces to find the one that had the greatest distinction between the brown of the soil and the brown color of the iron and manganese paints.
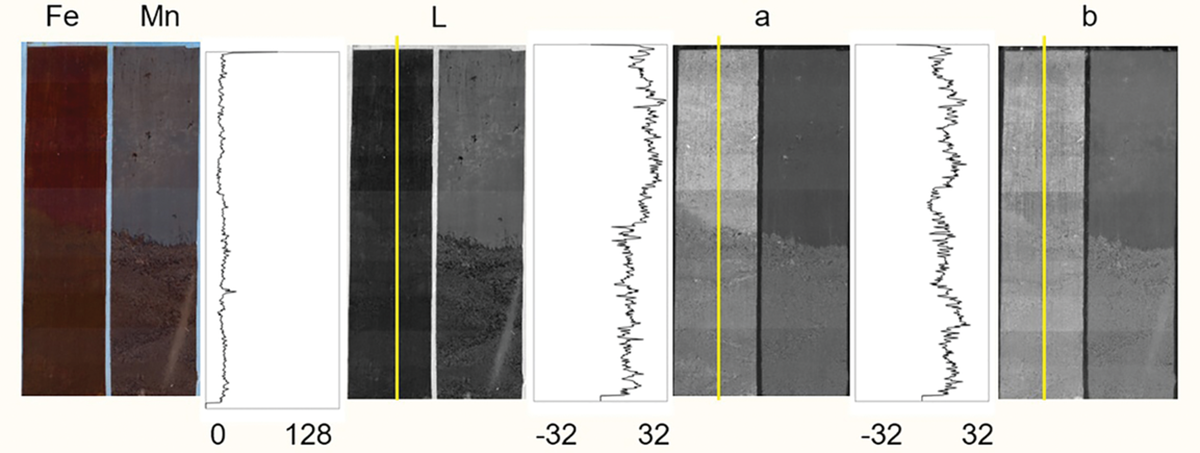
Here is an example of iron (Fe) and manganese (Mn) images from the laboratory setup on the left. The top sections show the oxide paint while the bottom sections are areas with the paint removed. The greyscale images on the right show channels in the CIE L*a*b* color space. Image by Thorsten Knappenberger.
Essentially, Knappenberger found that by using the CIE L*a*b* color space, he could collect data on the exact L*a*b* values where the iron had been reduced and soil showed through, as well as the value for the intact iron paint. He then used image analysis to convert these into a binary value, either “reduced” or “not reduced,” depending on whether the pixel’s L*a*b* value was closer to the baseline paint or soil values. For manganese, using saturation from the hue, saturation, value (HSV) color space worked best.
“This worked well after we found the right color value,” Knappenberger says. “There are definitely opportunities here to use supervised machine learning to train an algorithm. You could show it, ‘this is reduced, this area is not reduced,’ and it could find the best color channel for your soil, your system.”
Once Knappenberger created these binary images, it was time to see iron and manganese reduction on an hour-by-hour basis. In fact, they found that manganese-reducing conditions were present after about eight hours at the earliest in their mesocosm. At the latest, the team found that 30% reduction and removal of paint was reached after 13.4 days at the very latest. Knappenberger even created an animation showing reduction status by the hour.
“This study is so important for wetland protection,” says LeFevre, who is currently studying for her master’s in biosystems engineering at Auburn. “The quicker and more efficiently we can identify these wetlands, the better. And the IRIS method traditionally uses a ton of PVC—our method could be more sustainable.”
The experiment serves as a proof-of-concept, showing that it is possible to collect hourly reduction-oxidation data in the laboratory. In the future, the technique could be adapted to use low-cost cameras or could be taken to the field though researchers in different areas will have to calibrate their own color models to differentiate reduced areas from the paint colors applied to clear plastic tubes.
The new, adapted method gives us the first real-time view of the dynamics of iron and manganese reduction in wetland soil. It could aid researchers in better identifying how environmental conditions like temperature, microbial communities, and soil carbon affect these dynamics. It’s just one more tool in a wetland researcher’s toolkit to help us better understand and protect our invaluable wetlands and all the ecosystem services they provide.
Dig deeper
Check out the new method in Agricultural & Environmental Letters, “Camera Illustration of Indicator of Reduction in Soils (IRIS) Reduction Dynamics,” here: https://doi.org/10.1002/ael2.20051. Also of interest are:
- “Indicator of Reduction in Soil (IRIS): Evaluation of a New Approach for Assessing Reduced Conditions in Soil,” by Castenson & Rabenhorst, 2006 (https://doi.org/10.2136/sssaj2005.0130)
- “Macro and Microscopic Visual Imaging Tools to Investigate Metal Reducing Bacteria in Soils,” by Scott et al., 2021 (https://doi.org/10.1002/saj2.20171).
- “Development and Application of the Hydric Soil Technical Standard,” by Berkowitz et al., 2021 (https://doi.org/10.1002/saj2.20202)
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.