Elemental sulfur performance depends on source, soil type, and application timing

Elemental sulfur fertilizers don’t all behave the same. A recent study reveals how source, soil type, and especially temperature patterns after application dramatically influence when sulfur actually becomes available to crops. By uncovering why some products release sulfur too slowly while others meet early-season demand, the findings offer practical insights CCAs and their growers can use to better time applications and avoid hidden yield losses.
Earn 0.5 CEUs in Nutrient Management by reading the article and taking the quiz.
Declining atmospheric sulfur (S) deposition together with increasing crop yield potential has increased S removal, contributing to greater risk of S deficiency across U.S. cropping systems. Elemental S is a concentrated S source that can supply S over a growing season. Crops take up S primarily as sulfate; therefore, elemental S requires microbial S oxidation before plant uptake can occur. Because S oxidation is driven by soil microorganisms and environmental conditions, timing of plant-available S depends on source, soil type, and soil temperature following application.
Study overview
In Oregon, S oxidation was evaluated in Quincy fine loamy sand (mixed, mesic Xeric Torripsamment) and Walla Walla silt loam (coarse-silty, mixed, superactive, mesic Typic Haploxeroll) using laboratory incubation temperature regimes designed to simulate fall/winter and spring soil temperature patterns based on long-term (30-year) climate averages for Hermiston (Quincy soil) and Pendleton (Walla Walla soil) over a five-month lab incubation.
Fertilizer sources included micronized elemental S (reference material), monoammonium phosphate combined with micronized elemental S, monoammonium phosphate combined with micronized elemental S and ammonium sulfate, bentonite-bound prilled elemental S, and monoammonium phosphate as a non-S control (Figure 1 and Table 1). All elemental S sources were applied at an equivalent target rate of 100 lb S/ac (50 mg S/kg S equivalent) to allow direct comparison of S oxidation among fertilizer sources.
| Sulfur source | Elemental S | Sulfate-S | Total S |
|---|---|---|---|
% | |||
| MAP + micronized elemental S | 16 | 0 | 21 |
| AMS + micronized elemental S | 5 | 5 | 10 |
| Micronized elemental S | 100 | 0 | 100 |
| Bentonite prill S | 90 | 0 | 90 |
Selected soil properties appear in Table 2 to provide context for interpretation of soil effects.
| Parameter | Walla Walla silt loam | Quincy sandy loam |
|---|---|---|
| Organic matter (%) | 2.4 | 0.9 |
| pH | 6.1 | 8.1 |
| SO4–S (ppm) | 2.4 | 3.2 |
| Total S (ppm) | 80 | 136 |
Elemental S source and S oxidation potential
Across soils and temperature regimes, bentonite-bound prilled elemental S showed limited S oxidation during the evaluation period across soils and temperature regimes (Figure 2). Aggregation within prills reduced exposed elemental surface area until physical breakdown occurred. Prilled elemental S functioned more as a longer-term S source than a contributor to immediate sulfate availability. Earlier application timing or pairing with sulfate-S may help support early crop uptake where needed.
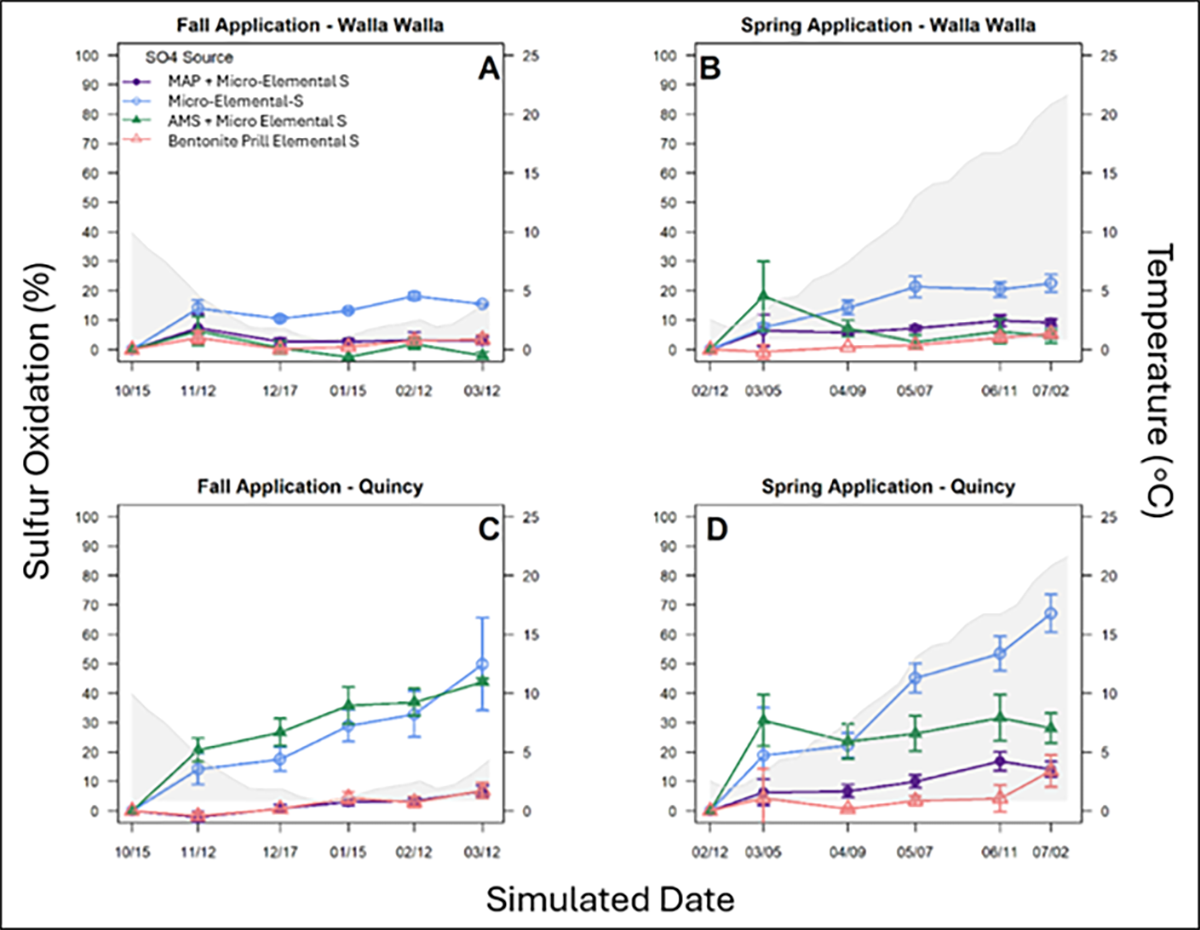
Monoammonium phosphate combined with micronized elemental S also showed limited S oxidation compared with non-granulated micronized elemental S used as a reference material (Figure 2). Granulation of micronized elemental S with MAP likely reduced exposed elemental surface area, resulting in slower S oxidation. Monoammonium phosphate + micronized elemental S contributed to S supply over time rather than rapid sulfate availability. Earlier application timing or inclusion of sulfate-S may help with early plant S nutrition needs.
Addition of ammonium sulfate supplied immediate sulfate-S while elemental S continued undergoing S oxidation over time (Figure 2). Co-granulated materials containing sulfate-S provided both early sulfate availability and continued S supply over a growing season.
Soil pH and texture effects on S oxidation
Elemental S oxidized more rapidly in Quincy fine loamy sand and more slowly in Walla Walla silt loam across evaluated fertilizer sources (Figure 2). Differences between soils indicate that soil type influenced elemental S oxidation processes. Quincy soil represented an alkaline, coarse-textured soil while Walla Walla soil represented a slightly acidic, finer-textured soil, suggesting that both soil acidity/alkalinity and soil physical conditions may contribute to differences in S oxidation. Acidic soil conditions corresponded with slower S oxidation although soil texture and associated aeration may also influence biological processes involved in S oxidation. Because soil pH and texture differed simultaneously between evaluated soils, independent effects of acidity/alkalinity and texture cannot be separated within this study. Additional research comparing soils with similar pH but differing texture, and soils with similar texture but differing pH, would help clarify which factor contributes more strongly to S oxidation responses.
Elemental S application timing
Application timing influenced S oxidation across temperature regimes (Figure 2). Elemental S applied under simulated fall conditions experienced approximately 40–50 °F soil temperatures for one to two months following application, followed by roughly three months of near-freezing temperatures through simulated winter temperatures. Measurable S oxidation occurred during the initial weeks after fall application, but subsequent near-freezing soil temperatures corresponded with slower S oxidation and delayed sulfate formation into later portions of the evaluation period. Soil temperatures approaching freezing reduced S oxidation rates and delayed sulfate availability.
Soil temperatures approaching freezing reduced S oxidation rates and delayed sulfate availability.
Elemental S applied under simulated spring conditions began at cooler soil temperatures (approximately mid-30s °F on February 12) but experienced steadily increasing temperatures through simulated March, April, and May conditions, reaching approximately 70 °F (Figure 2). Increasing soil temperatures into biologically active ranges corresponded with continued S oxidation over time and resulted in earlier sulfate availability compared with the fall regime. Soil temperatures increasing from near-freezing conditions in late winter to approximately 70 °F by early summer corresponded with faster S oxidation, even though initial temperatures were cooler at application.
Although fall application began under warmer soil temperatures than spring application, prolonged exposure to near-freezing conditions corresponded with reduced S oxidation later in the evaluation period. Temperature patterns following application influenced timing of S oxidation more strongly than starting temperature alone.
Summary
Elemental S fertilizers differed in S oxidation depending on fertilizer source, soil conditions, and temperature patterns following application. Faster S oxidation occurred where soils were alkaline and well aerated and where temperatures increased after application. Matching fertilizer source and application timing with soil conditions may help improve predictability of S availability.
This article is a contribution from the the Western Region Nutrient Management and Water Quality Committee (WERA-103), which fosters research, education, and outreach on nutrient management to improve crop efficiency, soil health, and water quality in the Western U.S. and Canada. See all articles.
Self-study CEU quiz
Earn 0.5 CEUs in Nutrient Management by taking the quiz for the article. For your convenience, the quiz is printed below. The CEU can be purchased individually, or you can access as part of your Online Classroom Subscription.
1. Which factor most directly determines when elemental S becomes plant available following application?
a. Total S concentration in the fertilizer.
b. S oxidation influenced by fertilizer source, soil type, and temperature.
c. Application rate alone.
d. Soil potassium levels.
2. Which fertilizer source provided immediate plant-available sulfate in the study?
a. Bentonite-bound prilled elemental S.
b. Monoammonium phosphate + micronized elemental S.
c. Co-granulated materials containing ammonium sulfate.
d. Micronized elemental S alone.
3. Compared with Quincy fine loamy sand, Walla Walla silt loam showed
a. faster S oxidation due to higher organic matter.
b. similar S oxidation regardless of soil conditions.
c. immediate sulfate availability from elemental S.
d. slower S oxidation potentially associated with slightly acidic conditions and fine soil texture.
4. Based on the simulated temperature regimes, which statement best describes application timing effects?
a. Temperature patterns after application influenced S oxidation more strongly than starting temperature.
b. Starting soil temperature determined S oxidation more than later conditions.
c. Fall application always resulted in faster S oxidation.
d. Spring applications prevented microbial S oxidation.
5. Elemental S can be taken up directly by crops without microbial S oxidation.
a. True.
b. False.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.