Considering biosolids as a sulfur nutrient source

Understanding the sulfur (S) value of biosolids produced by various processing methods is important for growers who routinely apply biosolids to crop production fields and rely on the S to support optimal plant growth and crop yields. It can be challenging to determine though since S is typically bound in organic and mineral compounds that are either mineralized or dissolved over a period of several months or longer after coming in contact with the soil. The goal of the research described in this article was to determine how processing method and biosolid properties influence S availability and fertilizer value through lab incubation.
This article was prepared as a contribution of the Western Region Nutrient Management Coordinating Committee (WERA-103).
Understanding the sulfur (S) value of biosolids produced by various processing methods is important for growers who routinely apply biosolids to crop production fields and rely on the S to support optimal plant growth and crop yields. Sulfur is taken up by plants in the form of sulfate (SO4–2). Sulfate is a moderately mobile nutrient that leaches out of the soil slowly over time and needs to be replaced with S-containing nutrient sources to meet the continued S needs of agronomic crops. Determining the S fertilizer value of waste materials like biosolids can be a challenge as S is typically bound in organic and mineral compounds that are either mineralized or dissolved over a period of several months or longer after coming in contact with the soil. As processing information of biosolids can be difficult for growers to access, chemical characteristics may be a good approximation of plant-available S in biosolid products. Our project goal was to determine how processing method and biosolid properties influence S availability and fertilizer value through lab incubation.
Approach
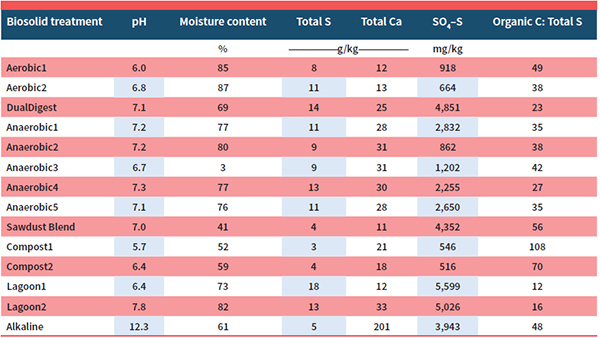
Fourteen biosolid materials were selected for the trial, representing the biosolid processes of aerobic digestion (n = 2), anaerobic digestion (n = 5), dual digestion (n = 1), composting (n = 2), lagoon storage (n = 2), sawdust blending (n = 1), and alkaline stabilization (n = 1). Biosolids were air-dried, ground, and added to a Walla Walla silt loam soil at a rate of 100 mg S/kg soil, which is roughly equivalent to 200 lb S/ac. The treated soils were incubated for 12 weeks to allow for S mineralization, oxidation, and dissolution processes to convert S to sulfate-S (SO4–S). The treated soils were extracted for SO4–S over the course of the incubation. Plant-available S was calculated based on the proportion of total S content that was released from the biosolids as SO4–S over the course of the incubation. The chemical characteristics of biosolids are listed in Table 1.

Mean plant-available S was greatest for the alkaline-stabilized biosolids (52%), followed by anaerobically digested biosolids (40%), sawdust-blended biosolids (37%), lagoon slurry (36%), dual-digestion biosolids (31%), aerobically digested biosolids (15%), and composted biosolids (4%) (Table 2).
Anaerobic vs. Aerobic Digestion
The range in mean plant-available S for the two aerobically digested biosolids (11–19%) was lower than the mean plant-available S range for the five anaerobically digested biosolids (25–37%). The primary differences occurred due to S mineralization rate rather than initial SO4 concentration (Table 2). Organic C:total S and total S content was similar among biosolids from the two digestion methods. The higher S mineralization rates for the anaerobic-digested biosolids compared with the aerobic-digested biosolids is likely caused in part by the mineralization of organic S during the aerobic processing. Aerobic conditions are far more conducive for S mineralization than anaerobic conditions. Another notable difference between the two processing methods is biosolid Ca concentration with anaerobically digested biosolids consistently containing more than twice the amount of total Ca as the aerobically digested biosolids. As mentioned above, the presence of Ca may indicate greater S availability from the dissolution of CaSO4 compounds. In either case, our findings suggest that aerobic digestion may significantly reduce S availability compared with anaerobic digestion.
Composting Biosolids
While the composted materials originated from different biosolids (sewage sludge vs. aerobically digested biosolids) and had different feedstocks (wood residue/yard waste vs. septage/yard waste), the S immobilization patterns were very similar among the two materials (Table 2). The composted biosolids were also the only biosolids to immobilize S during the 12-week incubation. Sulfur immobilization in the composted biosolids treatment was most likely driven by C:S, as these materials had the highest C:S of all of the biosolids evaluated in this study (70:1 and 108:1). With that said, the release of SO4–S immediately following application counteracted SO4–S losses from S immobilization with a very small albeit positive mean range in plant-available S for the two composted materials (2.8–5.6%). Composting biosolids may greatly restrict or even prevent the release of plant-available S although evaluation of additional composted biosolids may be needed to verify this finding.
Storing Biosolids in Lagoons
The two lagoon slurries evaluated produced percent plant-available S (26–46%) that was either similar to or significantly greater than percent plant-available S for anaerobically digested biosolids (27–37%). With the Lagoon2 treatment having roughly twice the S mineralization rate and almost three times the biosolid Ca concentration as the Lagoon1 treatment (Table 2), our findings would suggest that variations in S availability were likely caused by differences in CaSO4 concentration. Storing biosolids in a lagoon may either maintain or increase S availability compared with anaerobically digested biosolids, depending on the Ca concentration as CaSO4 in the lagoon. Further evaluation with a larger number of lagoon slurries is needed to validate these findings.
Stabilizing Biosolids with Alkaline Materials
Our findings would suggest that alkaline stabilization greatly increased S availability compared with anaerobic digestion. As described above, the cause for high S availability in alkaline-stabilized biosolids is more likely a result of CaSO4 dissolution than actual mineralization of organic S compounds. Highly reactive liming materials, like calcium oxide or calcium hydroxide, are added to biosolids to increase pH above 12.0 with the goal of limiting biological activity. Calcium oxide is highly reactive, leaving behind Ca compounds that may precipitate out with SO4–S to form a large quantity of CaSO4. Further evaluation of other stabilized alkaline biosolids for S availability and presence of CaSO4 compounds is needed to explore this material further as a S source for plants.
pH, Calcium: Sulfur Ratio, and Calcium Concentration Effect on S Availability
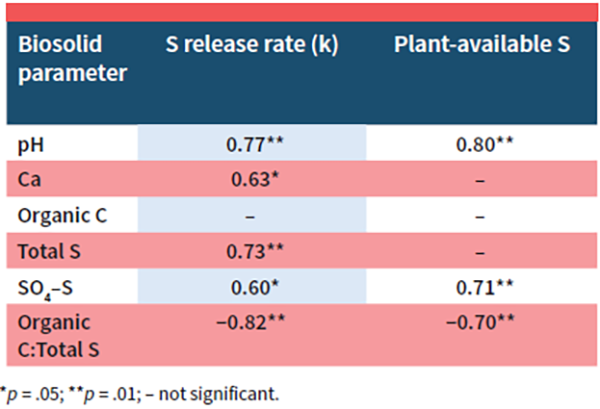
Biosolid pH and organic C:total S ratio as well as total calcium (Ca), total S, and SO4–S concentrations were highly correlated to either S release rate (k), plant-available S, or both (Table 3). Biosolid organic C:total S ratio is likely connected to the S mineralization of organic S compounds while Ca and pH correlations to S availability could be connected to gypsum (CaSO4) dissolution and the oxidation of hydrogen sulfide compounds. The correlations proposed here may need to be further investigated and verified on additional soil types and under field conditions prior to grower adoption.
Conclusion
Results showed that biosolid S properties, S mineralization dynamics, and plant-available S were significantly impacted by processing method, suggesting that the method should be considered when providing S availability estimates for biosolids. Biosolid properties correlating to S mineralization rate and plant-available S were identified. Biosolid Ca, S, SO4–S, C:S, and pH correlated significantly to S mineralization rate while biosolid pH, SO4–S, and C:S were highly correlated to plant-available S. The correlations proposed here may need to be further investigated on additional soil types and under field conditions prior to verification and grower adoption.
Dig deeper
View the full article, “Biosolids Processing Effect on Sulfur Plant Availability,” published in the Soil Science Society of America Journal at https://doi.org/10.1002/saj2.20379.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.









