Testing in Washington identifies widespread postemergence herbicide resistance in annual grasses

Use of herbicides that are ineffective cost growers in the short and long term. Herbicide resistance screening can help growers and agronomists by allowing them to understand which herbicides are no longer an option. This article reports on testing in Washington.
Herbicide resistance in the dryland wheat-producing regions of Washington is of critical concern due to long-term and repeated use of a very limited number of postemergence herbicides. Reduced- and minimum-disturbance cropping systems have created a heavy reliance on postemergence herbicides for grass weed control due to limited options for mechanical control, economically feasible rotational crops that also have effective and different management options, and availability and opportunity to use effective preemergence herbicides. In addition, climate has a significant impact on the effectiveness of preemergence herbicides.
The dryland wheat production regions of Washington are spread across a significant rainfall gradient, and more than half of the production region receives less than 15 inches of rainfall per season—some less than 7 (Schillinger et al., 2006). Many growers complain about inconsistent or limited effectiveness of preemergence herbicides as these herbicides require rain for activation. The large degree of climate heterogeneity contributes to differences in crop rotation and problematic annual grass weeds found throughout the eastern half of the state (Schillinger et al., 2006). Italian ryegrass, a spring annual, infests areas of relatively high annual rainfall (>18 inches annually) while downy brome, a winter annual, plagues areas with relatively less rainfall.
ACCase- and ALS-Inhibiting Herbicides
The most common postemergence herbicides used in dryland wheat production in Washington belong two herbicide mode of actions; the acetyl CoA carboxylase (ACCase; WSSA Group 1) inhibitors and the acetolactate synthase (ALS; WSSA Group 2) inhibitors. Glyphosate, an inhibitor of 5-Enolpyruvylshikimate-3-phosphate synthase (ESPS; WSSA Group 9), is commonly used in the fallow or crop transition period for nonselective postemergence weed control. Glyphosate use in crops is also increasing—canola acreage is increasing in eastern Washington as growers plant Roundup Ready canola varieties in annual-cropped areas.

ACCase-inhibiting herbicides are used to control grass weeds in dicot crops and cereal or rice crops with the help of crop safeners (Wenger et al., 2012). The ACCase inhibitors include three chemical families: the cyclohexanediones (DIM), aryloxyphenoxy-propionate (FOP), and phenylpyrazolines (DEN). Many herbicides in the ACCase inhibitors mode of action have been available for use for more than 20 years. Quizalofop-p-ethyl as Assure II (USEPA Reg. No. 5481-646) has been labeled since 1990, clethodim has been available since 1993, clodinafop as Discover NG (USEPA Reg. No. 100-909) and fenoxaprop as Puma 1EC (USEPA Reg. No. 264-666) since 2000, and the latest to be registered was pinoxaden as Axial in 2005 (USEPA, 2020).
ALS inhibitors have been used since the mid-1970s for control of both grass and broadleaf weeds, depending on the specific active ingredient, in a variety of crops (Levitt, 1978). Sulfosulfuron, as Maverick or Outrider (USEPA Reg. No. 59639-223), has been used since 1999, mesosulfuron as Osprey (USEPA Reg. No. 264-802) and propoxycarbazone as Olympus (USEPA Reg. No. 264-809) since 2004, and pyroxsulam at Powerflex (USEPA Reg. No. 62719-569) labeled for use in 2008 (USEPA, 2020). Imazamox was first labeled in 1997 at Raptor (USEPA Reg. No. 241-379) for use in broadleaf crops and then later became available in wheat with the commercialization of the Clearfield system in 2003 (Ball & Peterson, 2007; USEPA, 2020; Tan et al., 2005).
Testing for Herbicide Resistance
Herbicide resistance can manifest in three distinct syndromes: single, where the weed is only resistant to a single herbicide or herbicides in the same chemical family; cross, where the weed is resistant to herbicides in different herbicide chemical families within a mode of action; and multiple resistance, where the weed is resistant to herbicides in different modes of action. ACCase and ALS inhibitors are frequently applied every other year, if not every year, so risk for selection of resistance is high. There is considerable concern among growers and industry professionals about increasing resistance development among weed populations in Washington to both ACCase inhibitors and ALS inhibitors, particularly to manage downy brome and Italian ryegrass.
As a consequence, the Washington State University (WSU) weed science program began accepting mature weed seed from growers, industry professionals, and extension agents in Washington with suspected herbicide resistance in 2013. As of Jan. 1, 2020, the most common weed species submitted to the herbicide resistance screening program are Italian ryegrass and downy brome with suspected resistance to ACCase inhibitors, ALS inhibitors, and in the case of downy brome, glyphosate (see the Herbicide-Resistant Weeds Map at https://bit.ly/3joGYka for a geographical perspective on the cases of herbicide resistance in eastern Washington).
Testing for herbicide resistance is laborious, primarily because the simplest method is to spray the weeds. Submitted seed samples are cleaned by hand and stored in a dry area at room temperature. Plants are grown in a greenhouse ideally during the winter months after sample receipt. In some cases, biotypes are not screened until 9 to 12 months after receipt due to seed dormancy or immaturity. Herbicide rates are based on the maximum labeled rate for application in wheat in Washington, and adjuvants are included according to label requirements or field practice (Table 1). Two days after treatment, plants are cut down to an inch in height to monitor regrowth and reduce biomass variation among plants. The experiments are rated on a regular schedule, and at 21 days after treatment, aboveground leaves are harvested and weighed. A report is generated for each sample and sent to the sample submitter.
Table 1. Herbicide treatments, rate, adjuvants, and grass species tested for each treatment to determine if resistance was present in individual biotypes
| Treatment | Rate | ||||
|---|---|---|---|---|---|
| Trade name | a.i. | Mode of action | Field rate | lb a.i./ac | Grass species tested |
Assure II COC | quizalofop | 1 | 12 fl oz/ac 1% v/v | 0.0825 | Downy brome & Italian ryegrass |
| Axial XL | pinoxaden | 1 | 16.4 fl oz/ac | 0.0540 | Italian ryegrass |
Discover NG MSO | clodinafop | 1 | 16 fl oz/ac 0.25% v/v | 0.0625 | Italian ryegrass |
Clethodim 2 EC COC | clethodim | 1 | 16 fl oz/ac 0.50% v/v | 0.2500 | Downy brome & Italian ryegrass |
Beyond NIS UAN | imazamox | 2 | 6 fl oz/ac 0.25% v/v 2 qt/ac | 0.0470 | Downy brome & Italian ryegrass |
Olympus NIS UAN | propoxycarbazone | 2 | 0.90 oz/ac 0.25% v/v 2 qt/ac | 0.0394 | Downy brome |
Osprey NIS UAN | mesosulfuron | 2 | 4.75 oz/ac 0.25% v/v 2 qt/ac | 0.0134 | Downy brome & Italian ryegrass |
Outrider NIS UAN | sulfosulfuron | 2 | 0.66 oz/ac 0.25% v/v 2 qt/ac | 0.0310 | Downy brome & Italian ryegrass |
Powerflex HL NIS UAN | pyroxsulam | 2 | 2 oz/ac 0.25% v/v 2 qt/ac | 0.0164 | Downy brome & Italian ryegrass |
RT3 AMS | glyphosate | 9 | 28 fl oz/ac 8 lb/100 gal | 0.9800 | Downy brome & Italian ryegrass |
Italian Ryegrass
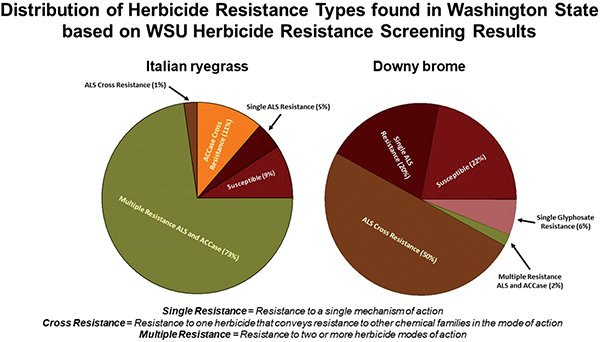
The 44 Italian ryegrass biotypes tested vary in their pattern of herbicide resistance (Figure 1). The majority, 73%, of the total Italian ryegrass biotypes submitted presented multiple herbicide resistance to both ACCase- and ALS-inhibiting herbicides. Multiple resistance presented in different patterns with eight biotypes resistant to all herbicides applied, 16 biotypes resistant to all herbicides applied except for one to two active ingredients, and eight biotypes resistant to one to three ACCase inhibitors and one to two ALS inhibitors. There were 10 Italian ryegrass biotypes with multiple resistance, but clethodim was still a viable option for control. Cross resistance was found in 14% of the samples submitted with five biotypes cross-resistant to ACCase inhibitors and one biotype cross-resistant to ALS inhibitors. Resistance to a single herbicide occurred for only 4% of the Italian ryegrasses submitted with resistance to pyroxsulam in two biotypes. There were four biotypes submitted (9%) that did not have resistance to any herbicide screened.
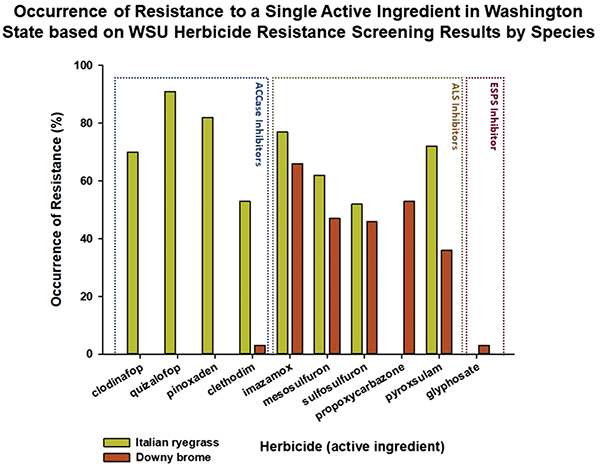
The occurrence of resistance in the suspected resistant Italian ryegrasses submitted to a single herbicide active ingredient in the ACCase inhibitor mode of action was 91% for quizalofop, 70% for clodinafop, 82% for pinoxaden, and 53% for clethodim. For ALS inhibitors, the occurrence of resistance in Italian ryegrass biotypes submitted was 77% for imazamox, 62% for mesosulfuron, 52% for sulfosulfuron, and 72% for pyroxsulam (Figure 2). No Italian ryegrass biotypes have been submitted with suspected resistance to glyphosate as on January 2020, and glyphosate resistance has not been found in any Italian ryegrass biotype tested in Washington.
Downy Brome
A total of 50 downy brome samples have been submitted and screened with herbicides of suspected resistance. The majority, 94%, of the downy brome biotypes submitted had suspected resistance to ALS inhibitors. Of those, 2% had multiple resistance to both ALS inhibitors and an ACCase inhibitor, 52% were found with cross resistance to multiple chemical families of ALS inhibitors, 20% were found with single resistance to one ALS-inhibiting chemical family, and 22% were susceptible to all herbicides screened (Figure 1). Clethodim resistance occurred in 2% of the total downy brome biotypes submitted for resistance screening. Cross-resistance patterns differed by biotype with nine biotypes having cross resistance to all ALS-inhibiting chemical families tested and the remainder having resistance to two to three different ALS chemical families. The occurrence of resistance to a single herbicide in the ALS-inhibiting family in downy brome was 66% for imazamox, 47% for mesosulfuron, 46% for sulfosulfuron, 53% for propoxycarbazone, and 35% for pyroxsulam (Figure 2). Currently, three biotypes have been submitted with resistance to glyphosate ranging from 88 to 165 times more resistant than the susceptible biotype. These three biotypes do not have cross resistance to any other herbicide mode of action.

In total, 84% of the grass weed biotypes submitted to the WSU herbicide resistance screening program were resistant to either ACCase inhibitors, ALS inhibitors, or both. All downy brome samples (three biotypes) submitted with suspected glyphosate resistance were confirmed to have glyphosate resistance. Interestingly, 24% of the suspected resistant biotypes submitted were found not to be resistant to the suspected herbicide. Of those misidentified biotypes, 43% were susceptible biotypes to ACCase, ALS, and glyphosate. Other factors such as high weed pressure/populations, late application timings, non-lethal application rates, inadequate spray coverage, and late germination could all contribute to the number of incorrect identifications of herbicide resistance. Monitoring of weed populations in a field should be a priority because detecting the correct herbicide resistance is the first step in making adequate management plans to mitigate the resistant population's impact (Soteres et al., 2015).
Short- and Long-Term Costs of Ineffective Herbicides
Herbicide resistance screening can help growers and agronomists by allowing them to understand which herbicides are no longer an option. Use of herbicides that are ineffective cost growers in the short and long term. Short-term costs include the expense of the initial non-lethal herbicide and application (fuel, equipment depreciation, equipment hiring costs, etc.), loss of income due to commodity dockages, and additional herbicides and mechanical management options later in the season due to the initial failure. Long-term costs include prolonged intensive weed management due to increased weed pressure from seed set(s) not prevented due to ineffective herbicide use. Prolonged intensive weed management could include increased costs due to increased cultural, mechanical, and chemical methods for an extended period, depending on weed population size and spread throughout the field. Example costs for cultural methods could include rotation crops that are not as economical, loss of valuable crop acres due to mowing non-harvestable acres, and higher seed prices for crops with herbicide traits. Mechanical inputs such as additional fuel, acquiring or modifying equipment not typically used, loss of government programs, and increased labor costs for things such as hand-weeding could be added. Lastly, chemical control method costs could increase because cheaper options are no longer available or are not effective. The longer a resistant weed population goes undetected, the greater the cost for managing it in the future.
References
Ball, D.A., & Peterson, C.J. (2007). Herbicide tolerance in imidazolinone-resistant wheat for weed management in the Pacific Northwest U.S.A. In H.T. Buck, J.E. Nisi, and N. Salomón (Eds.) Wheat production in stressed environments (pp. 243–250). Dordrecht, the Netherlands: Springer.
Heap, I. (2020). The international survey of resistant weeds. Retrieved from http://www.weedscience.org.
Levitt, G. (1978). Herbicidal sulfonamides. U.S. Patent4, 127, 405.
Llewellyn, R.S., Linder, R.K., Pannell, D.J., & Powles, S.B. (2002). Resistance and the herbicide resource: perceptions of Western Australian grain growers. Crop Protection21, 1067–1075.
Schillinger, W.F., Papendick, R.I., Guy, S.O., Rasmussen, P.E., & Kessel, C.V. (2006). Dryland cropping in the western United States. In G.A. Peterson (Ed.) Dryland agriculture (2nd ed., pp. 365–393). ASA, CSSA, and SSSA, Madison, WI.
Soteres, J.K., Thill, D.C., & Mallory-Smith, C.A. (2015). Sulfonylurea-resistant Russian thistle (Salsola iberica) survey in Washington State. Weed Technology8, 258–264.
Tan, S., Evans, R.R., Dahmer, M.L., Singh, B.K., & Shaner, D.L. (2005). Imidazolinone-tolerant crops: history, current status and future. Pest Management Science, 61, 246–257.
USEPA. 2020. EnviroAtlas. Pesticide product and label system. Retrieved from https://iaspub.epa.gov/apex/pesticides/f?p=PPLS:1
Wenger, J., Niderman, T., & Mathews, C. (2012). Acetyl-CoA carboxylase inhibitors. In Modern crop protection compounds (pp. 447–477). Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.