Assessing soil health: Soil carbon cycling and storage

The following article is the second in a five-part series on assessing soil health. It focuses on soil carbon cycling and storage. It is part of a larger Soil Science Society of America webinar series produced in partnership with The Soil Health Institute and sponsored by The Walton Family Foundation.
Carbon. Carbon. Carbon.” was the title of a memorable presentation given by a fellow soil scientist. Each slide reiterated carbon, and six slides later, the conclusion was carbon. Why is soil carbon so important? It is the food source for soil microbes, and microbial activity determines much of soil function. Soil microbes are directly responsible for nutrient cycling. However, soil microbes also indirectly affect air and water movement, nutrient cycling, and water filtration. For example, microbial activity, along with roots and weather, determine soil aggregation and structure development, and this soil structure creates the physical housing in which everything happens.
Welcome to the second installment of the “Assessing Soil Health” series, and you guessed it, this one is all about measurements of soil carbon cycling and soil carbon storage. This article will discuss soil carbon and soil organic matter and provide information on specific soil carbon cycling measurements. In this discussion, insights from general soil science literature and from the recent Soil Health Institute project titled, “The North American Project to Evaluate Soil Health Measurements (NAPESHM),” will be assimilated to help in understanding each measurement in the context of soil health management practices and soils.
The first item on the list, before delving into the fascinating ways we can measure soil carbon and microbial activity around cycling carbon, is to clearly discuss the terms of soil organic carbon and soil organic matter.
Soil Organic Matter
Soil organic matter (SOM) consists of all of the carbon-based compounds that were once in living organisms. It originates as plant and animal tissue, but then it is modified into other compounds by microbial processes during decomposition. Many commercial soil-testing labs measure SOM using a loss-on-ignition method that involves heating a soil sample in an oven to combust organic matter and weighing the sample before and after combustion to calculate SOM as a percentage by weight. The method is inaccurate because other mineral components can be combusted or dried (for example, water in gypsum).
Total Soil Carbon
Total soil carbon is accurately measured by a dry combustion method (Nelson & Sommers, 1996). In this method, soil is heated, and the CO2 evolution is measured. Because the method only measures CO2 evolved, it is more accurate than measuring a change in weight, like loss on ignition. The dry combustion method measures both organic and inorganic forms of C.
Soil Inorganic Carbon
Inorganic C in soil is primarily in the form of carbonates (CO32–) such as calcite (CaCO3) and dolomite (MgCO3). One way to check for the presence of carbonates is the fizz test; 10% HCl is dropped onto soil, and visible bubbles erupt from the sample. The bubbles are CO2 from the carbonates (CaCO3 + 2HCl = CO2 +Ca2++ 2Cl− + H2O). Inorganic carbon in soil can be measured quantitatively by measuring the production of CO2 in response to acid. In commercial labs, soils with a pH greater than 7.2 are usually marked for inorganic carbon testing.
Soil Organic Carbon
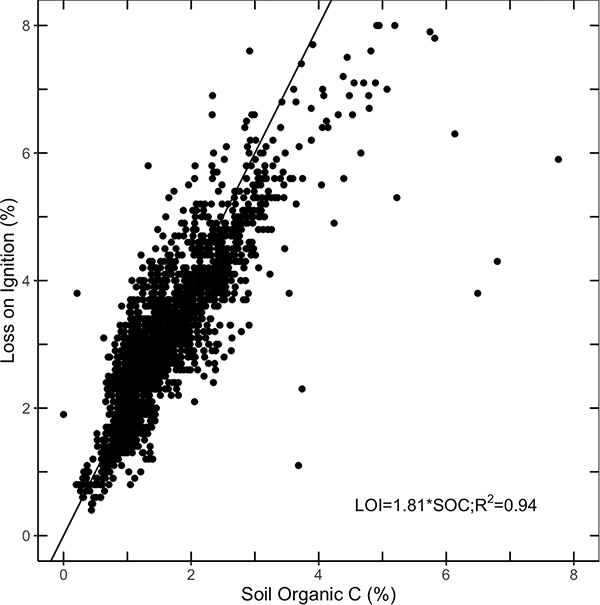
Organic carbon in soil is calculated as total carbon from dry combustion minus inorganic carbon. Soil organic carbon (SOC) and SOM are linearly related at roughly a ratio of 1:2 (SOC:SOM; Pribyl, 2010). Note in Figure 1, the relationship (1.8 slope) between SOC from dry combustion and SOM from loss on ignition in the NAPESHM data. The traditional conversion between organic carbon and organic matter is SOC × 1.72 = SOM. The multiplier is important to note because it is still the standard at many labs and throughout some organizations. To be consistent in comparisons over time, it is vital to check what method was used to measure your samples, especially if you have old data that reports SOM and you are updating to using SOC measured with dry combustion. The Soil Health Institute, NRCS (Stott, 2019), and others recommend that SOC be measured directly by dry combustion for assessing soil health (Nelson & Sommers, 1996).
Sampling and Measuring Soil Organic Carbon
Soil organic carbon is an important measurement to track and assess soil health, quantify soil carbon stocks, and understand nitrogen cycling. For tracking soil health, we recommend taking a composite soil sample of 15 to 20 subsamples per management unit from a 0- to 15-cm depth. For assessing carbon stocks, also pull a sample of known volume from 0 to 15 cm so that bulk density can also be measured. The bulk density measurement is used to convert organic carbon percent to a carbon stock (mass per unit area, e.g., lb or Mg of C per acre or ha). To document changes in carbon stock over time, also take a composite sample from 15 to 30 cm and a corresponding bulk density sample as well. Soil health samples should be taken generally at the same time of year (spring before planting is preferred) and near the same location. If interested in documenting carbon stock for a whole field or farm, there are recommendations for spatial sampling design that are beyond the scope of this article.
Other Measures of Soil Carbon: Pools and Enzymes
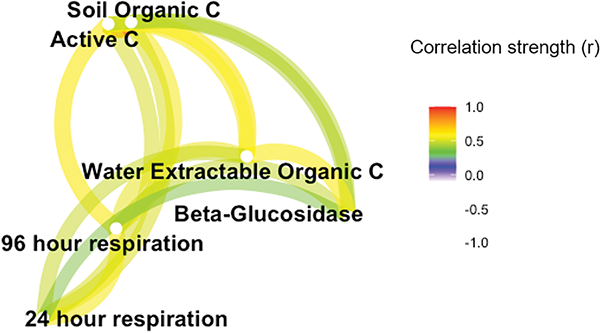
There are many other ways to measure and monitor changes in soil carbon cycling. At this time, the Soil Health Institute does not identify a preferred method. The usefulness and costs are discussed below, and the measurements are all moderately correlated with each other and SOC (Figure 2). Active carbon, water-extractable carbon, soil respiration, and beta-glucosidase enzyme activity are all soil carbon-cycling indicators (Table 1). These carbon-cycling indicators may have an advantage to SOC in that changes in soil are more speedily detected; hence, changes are more commonly detected within a few years of the management change. No obvious benefit to the grower or the CCA for measuring more than one of the carbon-cycling indicators has been identified by the Soil Health Institute to date.
Table 1. A general summary of carbon-cycling measurements and their meaning to soil health. More detail on methods can be found in Norris et al. (2020)
| Active C (permanganate oxidizable C; POXC) | Fraction of soil organic carbon that is oxidizable by a weak potassium permanganate solution, mg C kg–1 soil | Subsets the carbon pool using a defined chemical reaction. It is thought to reflect carbon compounds that are available to microbes. |
| Water-extractable organic C | Fraction of soil organic carbon extractable by water, mg C kg–1 soil | Represents a mix of carbon compounds. It is thought to reflect carbon compounds available for microbes to access. |
| Soil respiration (mineralizable C) | Flush of CO2 from rewetted soils during a short-term aerobic incubation, mg CO2–C kg–1 soil day−1 24-hour incubation (Haney test) 96-hour incubation (Cornell test) | A measure of microbial activity under standardized (anaerobic) conditions. Shown to be correlated to microbial biomass. |
| Beta (β) glucosidase | Soil enzyme activity responsible for the decomposition of plant cell walls, mg p-nitrophenol (pNP) kg–1 soil hour–1 | Indicates activity of an enzyme used in some C decomposition. We know what is being measured, no clear understanding of how to interpret. |
In the NAPESHM project, we measured the carbon storage and cycling indicators (Table 1) across 124 long-term research experiments in Canada, the USA, and Mexico. We found that all measurements were sensitive to changes in management except for increased diversity in crop rotation. Beta glucosidase, active carbon, and soil respiration most consistently responded to management changes (Table 2).
Table 2. Significant responses of soil carbon storage and cycling indicators to management from the data set in the North American Project to Evaluate Soil Health Measurements. “Yes” means that the indicator changed significantly (α = .05) as a result of the treatment listed
| Soil carbon storage and cycling indicators | Decreased tillage | Organic amendment | Increased crop number | Cover crops |
|---|---|---|---|---|
| Total organic C | Yes | Yes | No | No |
| Active carbon POXC | Yes | Yes | No | Yes |
| 24-hour respiration | Yes | Yes | No | Yes |
| 96-hour respiration | No | Yes | No | Yes |
| Water-extractable organic C | Yes | Yes | No | No |
| Beta glucosidase | Yes | Yes | No | Yes |
Summary
The evidence suggests that (1) many of the soil carbon-cycling tests are correlated, (2) the carbon-cycling tests may be likely to show statistical differences compared with SOC in timelines of less than five years, and (3) respiration, active carbon, and beta-glucosidase appear equally sensitive to management changes. Therefore, our preliminary recommendation for agronomists wanting to monitor soil health for a given soil under a given management is to measure soil carbon and select one measure of carbon cycling. The high correlation and similar direction response to management between soil carbon-cycling measures may mean that you can get a good idea of cycling from any of these measurements. Consistency in sampling and measurement is key to interpreting whether you are enhancing your soil health through changing management.

References
Haney, R.L., Brinton, W.F., & Evans, E. (2008). Soil CO2 respiration: comparison of chemical titration, CO2 IRGA analysis and the Solvita gel system. Renewable Agriculture and Food Systems, 23, 2, 171–176.
Nelson, D.W., & Sommers, L.E. (1996). Total carbon, organic carbon, and organic matter. In D.L. Sparks (Ed.), Methods of soil analysis: Part 3. Chemical methods ( 2nd ed., pp. 961–1010). Madison, WI: ASA and SSSA.
Norris, C.E., Bean, G.M., Cappellazzi, S.B., Cope, M., Greub, K.L.H., Liptzin, D., … Honeycutt, C.W. (2020). Introducing the North American Project to evaluate soil health measurements. Agronomy Journal, 112, 3195–3215. https://doi.org/10.1002/agj2.20234
Pribyl, D.W. (2010). A critical review of the conventional SOC to SOM conversion factor. Geoderma, 156, 75–83
Stott, D.E. (2019). Recommended soil health indicators and associated laboratory procedures (Soil Health Technical Note No. 450-03). Washington, DC: USDA-NRCS.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.