The potassium paradox
Clay type affects potassium availability

The type of clay you have in your soil could make a $10,000 or more difference in your potassium (K) availability. Groundbreaking research by North Dakota State University (NDSU) Extension Soil Specialist David Franzen and others reveals that a soil’s clay mineralogy is responsible for K fixation (tie-up in unavailable forms).
The type of clay you have in your soil could make a $10,000 or more difference in your potassium (K) availability. Groundbreaking research by North Dakota State University (NDSU) Extension Soil Specialist David Franzen and others reveals that a soil’s clay mineralogy is responsible for K fixation (tie-up in unavailable forms). For example, smectite clay particles hold onto K tightly, making it less available to plants (see sidebar).
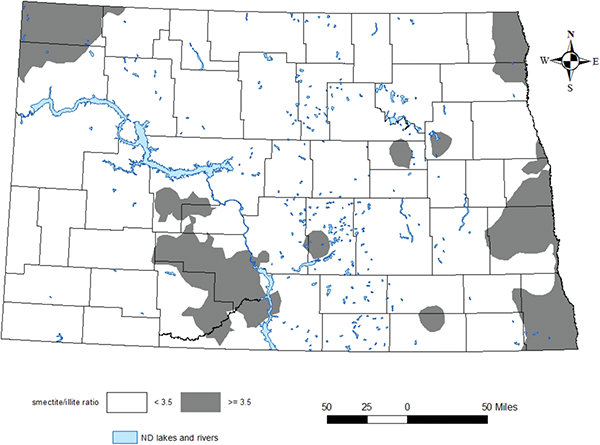
Minnesota research has similar findings in soybeans (see below). Specifically, a ratio of smectite/illite (clay components) above 3.5/1 required a soil test K critical level of about 200 ppm. The ratio of clay mineralogy correlates highly with corn yield response to K fertilizer.
In Franzen’s research, a smectite/illite ratio below 3.5/1 had a K critical level of 150 ppm. Stated another way, when the smectite-to-illite ratio was less than 3.5, the critical soil test K level was 150 ppm, 50 ppm lower than for a smectite/illite ratio above 3.5. High-illite clay soils had a critical soil test level 50 ppm lower than soils with less illite.
The Back Story
The devil is in the details when it comes to predicting K’s impact on corn yields in clay soils. What really matters is how much illite, smectite, and kaolinite comprise a soil’s clay particles. A soil’s clay composition and soil test K levels are further affected by soil moisture, tillage method, soil parent material, cation exchange capacity (CEC), and soil aeration.
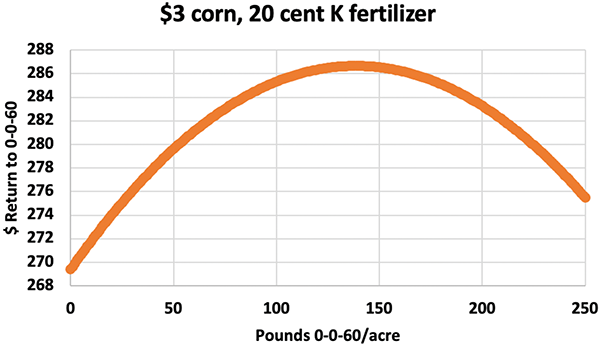
Until 2014, North Dakota’s K soil test values only predicted whether or not a site responded to K fertilizer about half the time. The soil K level that divided the responses lay at 150 ppm K, according to Franzen. He noticed that high-smectite clay soils had 20-30 bu/ac corn yield increases when 150-200 lb/ac of 0–0-60 was applied, yet in places where soils have more illite clays, yields did not respond to the same K rate applications. “We saw that wet, dry, or normal rainfalls did not influence corn yields on these higher-illite soils.”
Among six rates of applied 0–0-60, half the time, the highest 250 lb/ac rate decreased corn yields. “Wisconsin corn trials and Minnesota soybean trials were seeing this, too,” Franzen says.
From 1976 to 1994, Franzen was a fertilizer plant agronomist and manager in east-central Illinois. He noticed how local clay soils’ sensitivity to wet springs differed dramatically. “Illitic soils don’t fix themselves if abused by untimely field operations.” The illitic-clay-dominated soils (Elliot/Ashkum series) were unforgiving when tilled in wet springs while the Flanagan/Drummer series soils 10 miles south of there with the same topography could be worked earlier, even in wet springs, and were forgiving of ruts made in rainy springs. They have more smectite clays, he says, which are more forgiving.
It turns out that those soil behaviors during wet springs weren’t the only things influenced by the type of clay particles they had.
Four years of NDSU research found that the soil clay type affects K availability. North Dakota has three dominant clay types—smectite (shrinking–swelling 2/1 clays), illite (low shrinking–swelling 2/1 clays), and kaolinite (non-expanding 1/1 clays)—with a very small amount of chlorite (2/1/1 non-expanding clays).
Kaolinite’s K availability totally depends on CEC, but K availability of smectites and illites only partially depend on CEC. In illites, the K is released from surface charge and interlayer K whether the soil is wet or dry. Before this relationship came to light in Franzen’s four-year NDSU study, North Dakota K fertilizer recommendations were based on a soil test K critical level of 150 ppm. Above that K application level, a corn yield increase was regarded as unlikely.
Meanwhile, the state’s soil K values dropped rapidly over 30 years when corn–soybean rotations replaced wheat.
Franzen found that Activation Labs (Ancaster, ON, Canada) can do semi-quantitative X-ray diffraction clay speciation tests that document the relative composition of each individual clay type. Conventional soil-testing labs are unable to do this.
He has since updated NDSU K soil recommendations according to clay soil composition (illite/smectite ratio). North Dakota is the only state to have mapped its smectite, illite, kaolinite, and mica clay maps statewide due to taking samples for clay species and K-feldspar from multiple sites in every county in the state.
Soybeans Affected, Too
Various land grant university researchers are updating K recommendations, including Dan Kaiser, University of Minnesota Nutrient Management Specialist. “I bumped up Minnesota’s K rate recommendations in corn and soybeans and will probably do the same for other crops,” he says. He found soybean yield reductions from high K applications, similar to what NDSU’s Franzen observed in corn.
Kaiser doesn’t recommend more than 100 units/acre 0–0-60 or 60 lb/ac K2O before soybeans.
“I get CCA questions about K at every meeting these days,” he says. Being less costly than some other crop nutrients, K hadn’t gotten as much attention recently as, say, sulfur, in soybeans, Kaiser acknowledges.
Tri-state (Indiana, Michigan, and Ohio) K recommendations were recently updated and now use CEC to classify soils into two groups: sands or loams and clays.
What This Means
Both NDSU and the University of Minnesota have bumped up their K critical guidelines to 200 ppm for high smectite/illite ratio soils or medium- and fine-textured soils. The critical levels are also crop-specific—some have 200 ppm and others 120 or 130 ppm. In the upper Midwest, 20-40% more sites may require K application for the first time based on the new research, says John Breker, soil scientist and CCA, AgVise Laboratories, Northwood, ND. Breker was also part of Franzen’s soil K research as a graduate student.
“Our understanding of slowly available K and unavailable K has changed since the NDSU research and [Don] Sparks’ work at Delaware,” Breker says. “These non-exchangeable K and sometimes feldspar-K forms provide more K than we previously credited. It helps explain why we see nonresponsive sites on sandy soils even though soil test K might be low.”
Even sandy soils can have K-availability paradoxes when the sands’ parent material is K-feldspar. Potassium applications on lower-testing sandy Delaware soils triggered a few questions for Don Sparks at the University of Delaware in the early 1980s. Very sandy soils did not often respond to K applications. “This was ascribed to large quantities of K in feldspars in the soils’ sand fraction, which could be released during growing seasons,” Sparks says. “Crops also obtained K from the non-exchangeable K fraction as well as K in the subsoils, which had leached but was available for plant uptake (see the published research in the Soil Science Society of America Journal at https://bit.ly/2NRxd3F and https://bit.ly/3q41aul).
‘Show Me’ Missouri Clay Types
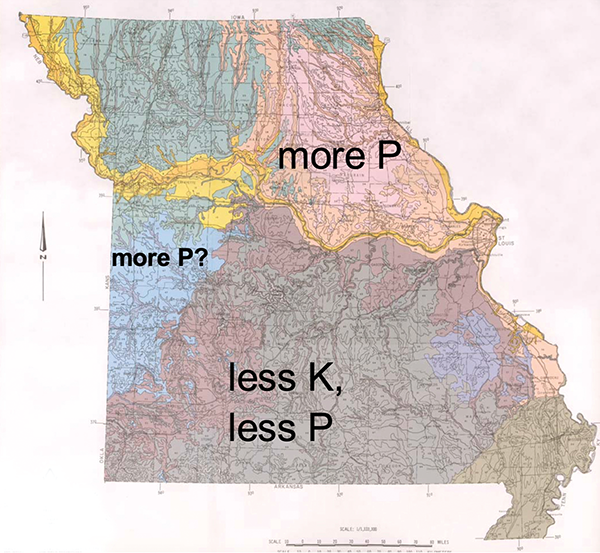
Peter Scharf, University of Missouri Plant Sciences Professor, also correlates soil K levels with clay minerals. “A soil’s K increase when fertilized depended on the region the soil came from,” he says, adding that clay mineralogy appears to be the reason (Figure 1).
“Our K hypothesis was that there would mainly be kaolinitic clays south of the Missouri River and smectite clays north of the river,” Scharf says. “The smectite clays would tie up more K in their interlayers, leading to a smaller soil test increase with added K fertilizer. Our results verified this. We found that Ozark soils (gray-brown and red-brown areas in Figure 1), where kaolinite dominates, required less K to raise soil test levels than soils in other regions. We expected the same for the Osage Plains region (blue area in Figure 1), but it wasn’t true; it turns out that soils in this region have a more mixed clay mineralogy. More K is applied in this region because it has a lot more row crops than the Ozarks.
“It’s not just the type of clay, it’s how much clay. We recommend more K for higher-clay soils,” as reflected in their CEC. This is supported by Scharf’s lab study (https://bit.ly/3ebBz0v) in which the soil test K levels of higher-clay soils increased less with the same amount of fertilizer. Some of the K is fixed by the clays in ways that don’t allow it to show up in a soil test.
What Has Not Changed about K Availability

When it comes to K availability, some things have not changed, according to North Dakota’s Franzen and Minnesota’s Kaiser:
- Soil moisture increases K’s soil movement, so response to K fertilizer is higher in dry years. Dry seasons may reduce K root uptake.
- Soil compaction and saturated soils impede K uptake because air is necessary for root respiration and K uptake. Root activity and subsequent K uptake decrease as soil becomes saturated and/or compacted. Soil oxygen levels are very low in those circumstances.
- Tillage system affects K availability with K becoming less available in no-till and ridge-till planting systems. The exact cause of this reduction isn’t known although research results point to restricted root growth to the surface of soil aggregates and not so much within the aggregate. Potassium fertilizers are concentrated in the top few inches, which limits the volume of roots available for their uptake. Banding, or deep banding, K may be ways to move K below the surface, according to Dan Kaiser.
- Soil temperatures of 60-80°F are ideal for maximum K uptake. Root activity, plant functions, and physiological processes increase as soil temperature increases, increasing K uptake.
- Potassium-deficient plants often have slow growth, poor drought resistance, weak stems, and are more susceptible to plant disease.
Potassium Basics
Available K is dissolved in soil water. This is the K form measured by soil tests’ so-called exchangeable K. It is held on the exchange sites of clay particles on their surface. Crops readily absorb the K dissolved in the soil water. As soon as the soil water’s K concentration drops, clay minerals release K attached to them. The K attached to the exchange sites of clay minerals is more readily available for plant growth than the K trapped between layers of clay minerals. Slowly available K varies in amount with the dominant type of clay and minerals that might have large amounts of clay, such as K-feldspars. The K is trapped between layers of clay minerals as non-exchangeable K or is released during natural weathering of the mineral. It therefore isn’t measured by routine soil-testing procedure but can be a reservoir for readily available K. The amount of slowly available K that can be used by plants during a single growing season can be large, as previously demonstrated by Don Sparks. Unavailable K makes up approximately 90–98% of total soil K, depending on soil type. Feldspar and mica minerals contain most of the K. Plants cannot use the K in this crystalline-insoluble form. Previously defined as unavailable K, we now know that mineral K found in mica and some feldspars act as slowly available K. The large amounts of mineral K in some soils make this a substantial source of K. North Dakota’s Franzen believes that the reason corn yield increases in the southeast part of this state only reached 30 bu/ac rather than the 80-bu responses seen in Iowa studies is that the soils in this North Dakota region contain up to 15% by weight K-feldspar.—Source: https://bit.ly/3b9lNRO
Meet the Clays
The types of soil clay components or mineralogy can strongly influence yield response to K fertilizers when your K soil test values range between 130 and 200 ppm. This was the conclusion of a four-year study from North Dakota State University (NDSU) on clay soil components’ impact on corn K requirements (see https://bit.ly/3kyEeSO).
Smectite Clays
Smectite clays shrink and swell as they become dry and wet. They temporarily fix or retain K when soil is dry and prevent K release as they shrink. This is a reversible process when the soil re-wets.
In order to offset smectites’ tight grip on soil K, high-smectite soil’s K critical level for corn should be raised from 150 to 200 ppm. High-smectite soils constitute hundreds of thousands of acres across the Midwest.
“Clay minerals are arranged similarly to flat pages in a book,” says NDSU Extension Soil Specialist David Franzen. “Negatively charged clay layers are held together by positively charged ions (K+), like glue.”
The trapped K, “between the pages,” or layers of clay minerals, isn’t measured by soil tests. It is largely unavailable to crops but can be a reservoir for readily available K. While some of it can be released for plant use during a growing season, it can also be fixed between clay layers and converted into slowly available K.
A significant portion of smectite clays’ K (figuratively “between the pages”) is exchangeable because there is greater surface area with charge in these clays compared with other clay types, Franzen explains. (Of course, other positive ions such as NH4+, Ca2+, Mg2+, Na+, and Al3+ also “swim” around depending on the soil pH.)
“Smectite layers, also arranged like pages of a book, allow much of the K between pages to become plant available during moist periods because of the wider spaces between individual mineral layers, allowing K to move from the interlayers to the soil solution where it’s available to plants. This K in the soil solution is immediately available for plant uptake,” Franzen says.
Three-sheet minerals joined by one from each side are called 2/1. Examples are smectites, mica, illite, and vermiculite groups. Ions of K+ reside on interlayer sites between the 2/1 layers as well as on the clay surfaces. Wetting and drying of these sheets (shrink–swell potential) destabilizes the soil surface and can produce cracks.
“Of the 2/1 clay minerals, smectite has much greater CEC than illite because smectite sheets are less attracted to each other compared with illites, resulting in a greater surface area of charge,” Franzen says.
Smectites include montmorillonite and beidellite.
North Dakota clay soils have relatively high K soil test values, but the readily available portion is much lower in smectitic clay-dominated soils when the soil is dry. There’s new evidence that potassium feldspars, potassium micas, and 2/1 clay parent materials influence how much K is available to crops. Yield response to applied K in K-feldspar soils is lower than in other clay parent materials.
“Smectite clays can trap or “fix” substantial amounts of potassium,” says Peter Scharf, University of Missouri Professor of Plant Sciences. “Illite and kaolinite clays are not built to do this.”
Illite Clays
Illite (non-expanding) clays do not fix K between layers when they become dry due to the large amount of K that holds the sheets together more tightly than for smectites. Because the clays are so saturated with K, the illites are able to release K from their interlayers regardless of the soil moisture content. So, in dry seasons with high-illite clay, soils provide more available K to crops than the same soils during wet seasons.
Because they don’t expand and contract, they’re unforgiving when rutted in wet springs.
“Illite mineral layers (like pages in a book) are closer together than smectite layers due to the high amount of K ions that hold the layers together,” Franzen explains. “This allows more K+ ion movement out of illite clays, making more K available. Very little of illite clays’ K ‘between the pages of the book’ is exchangeable; it is tightly bound between narrowly spaced layers, but it is so saturated with K that K is released regularly. This K is classified as non-exchangeable because it doesn’t reside on the clay surfaces and is not removed by standard soil test K extractants.”
Another term for illite is “hydrous mica,” meaning that the clay chemistry closely resembles the original mica mineral with interlayer saturation of potassium ions (K+), but with greater interlayer spacing from K loss through time. The 2/1 mineral layers of illite and mica are held together by potassium ions (K+) that have no water around them. The interlayer space is too narrow for other cations or water.
“When illite is in potassium-depleted soil, K+ begins to move out of the interlayers and into the surrounding soil where it can be taken up and used by plants,” Franzen says. “Other cations that are surrounded by water start moving from the adjacent soil into the illite interlayers, beginning at the edges where the K+ first was lost. This movement of hydrated cations into the interlayer pries opens the edges of the minerals, like pages in a book, allowing for more K+ to move out and more hydrated cations to move in. Using the book analogy, this process is like slowly lifting a page of a book. When illite clays are present in K-depleted soil, K+ ions exit the interlayers into the surrounding soil, becoming more available to plants. When K fertilizers are applied, K+ re-enters the clay layers.”
New NDSU research by Franzen finds that some of the K in illite interlayers is available to plants even though it is not accounted for in the standard soil test that measures only exchangeable K.
He noticed that high-smectite clay soils with 150–200 ppm soil test K, which supposedly was enough according to the old recommendations, had 20-30 bu/ac corn yield increases when up to 200 lb/ac of 0–0-60 K fertilizer was applied. Yet where soils had more illite clays, yields did not respond to the same K-rate applications even when the soil test K indicated they should.
“Our K recommendations at that time were only predictive half the time, among 29 North Dakota test sites using the dry soil test method,” Franzen says. Other K soil test methods were even less predictive.
The building blocks of mica, illite, smectite, and kaolinite are molecular sheets of silicon oxide and aluminum hydroxide. Minerals are classified by the arrangement of these sheets.
If one silicon oxide sheet adheres to one aluminum hydroxide sheet, the mineral is classified as 1/1. Kaolinite is an example of a 1/1 mineral. If an aluminum hydroxide sheet is sandwiched between two silicon oxide sheets, it is 2/1. Illites and smectites are two prominent 2/1 mineral groups.
The 1/1 and 2/1 minerals have a net negative charge, and therefore, attract positive ions (cations). In illite, the negatively charged 2/1 mineral sheets are held tightly together by potassium (K+) cations. In smectite, the 2/1 layers are farther apart than they are in illite and allow movement of K+ ions as well as ammonium (NH4+), calcium (Ca2+), magnesium (Mg2+), sodium (Na+), and aluminum (Al3+) between the layers.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.