Plant tissue testing: How precise Is my lab data?

The carpenter’s adage of “measure twice and cut once” is an important one in most things we do. But how precise is the tape measure capable of providing that information? In plant tissue analysis, we need to have an understanding of what is realistic with regard to the precision provided by laboratories and not attempt to interpret the results to a finer scale than what is actually achieved. Agricultural and environmental laboratories doing plant analysis are capable of great precision. However, some methods have good precision, and others are relatively less precise.
This article was prepared as a contribution of the Western Region Nutrient Management Coordinating Committee (WERA-103).
Data is the air needed by the lungs of our decision making. Bad data is like bad air—toxic to the organisms breathing it. It is important to measure what we do and to measure accurately and precisely.
Precise Data
Technologically advanced management of plants growing in soils can be greatly benefited by properly performed soil, water, and plant tissue analysis. But how accurate and precise does this data need to be? And, how well are our laboratories performing for each of the various tests?
Producing good data requires three critical steps, namely:
- Proper sampling
- Accurate and precise analysis
- Correct interpretation
For most of us, we can more easily control the first and last steps, but we often feel less control with regard to the analysis portion as we are typically depending on others for that. However, we can employ actions that allow us this control. This was outlined in a previous article in this magazine regarding the importance of having defensible recommendations, including laboratory credentials (see https://doi.org/10.1002/crso.20048). In addition to these actions, we can also understand the precision of data in general. We have open access to the collective data provided by the lab community. Herein, we provide an assessment of plant tissue analysis.
The Database
The Soil Science Society of America (SSSA) has the vast expertise of its more than 6,000 member scientists. The North American Proficiency Testing (NAPT) Program (https://www.naptprogram.org/) is operated as an activity of SSSA and governed by an oversight committee comprised of some of these experts as representatives of regional soil and plant analysis workgroups, scientific organizations, state/provincial departments of agriculture, and private and public laboratories. The NAPT program furnishes agricultural and environmental laboratories with quality control and quality assurance tools through quarterly blind and double-blind statistical evaluation of soil, plant, and water samples. These tools assist laboratories in generating accurate and precise analyses as well as leveraging their participation in assuring clientele and other consumers that their data meet high standards.
This valuable program, with the collective wisdom and expertise found in the credibility of SSSA, not only provides resources to laboratories, but also to consumers of the data they generate. The aggregated soil, water, and plant data generated by these laboratories are openly available at www.naptprogram.org/content/laboratory-results. A portion of this plant data was analyzed and presented at the recent Annual Meeting of SSSA (https://bit.ly/3u4ejGB). This plant data comes from three unique samples submitted to participant laboratories each quarter for them to test and then submit data for any or all of the 49 NAPT-accepted analytical methods. We examined a subsample from the library of plant tissue data available used for this evaluation of the first quarter (Q1) data from each year of 2011–2019.
Laboratories’ Report Card
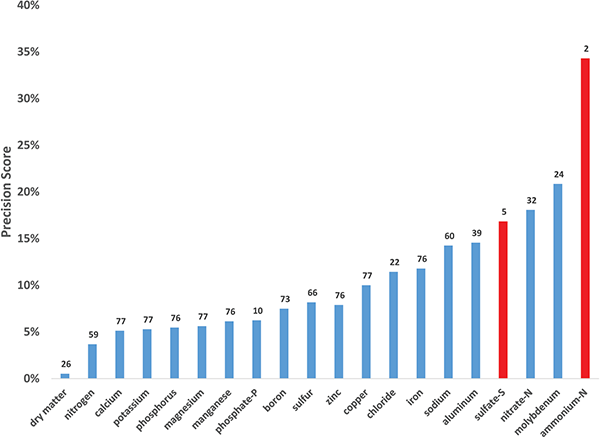
A measure of precision for each method was made by dividing the median absolute deviation (MAD) by the median for each of the 27 samples evaluated over these many years for each analyte. These precision scores were average = 11%, median = 8%, standard deviation = 10%, minimum = 4%, and maximum = 43%. The median data, combined across methods, is shown in Figure 1. These values give a sense of the precision of the data generated collectively across labs. There is much data to parse in this analysis, which will be the subject of a future, in-depth publication. However, there are some important preliminary points to glean for those using plant tissue data in their management.
Dry matter was the most precise method with a precision score of 0.5%. When combined across methods, the macronutrients were relatively precise with relative scores of nitrogen (4%), calcium (5%), potassium (5%), phosphorus (6%), magnesium (6%), and sulfur (8%). Some of the micronutrients lay within the top end of this range with manganese (6%), boron (8%), and zinc (8%). But, others were relatively less precise with copper (10%), chloride (11%), iron (12%), and molybdenum (21%). Sodium (14%) and aluminum (15%) were also less precise.
When analyzing some plant species, such as potato and cotton, the calibration data for nutrient management relies on extraction of a portion of the nutrient, rather than the total quantity (most commonly, nitrate-N and phosphate-P). The phosphate ion extraction was relatively precise (6%), but nitrate was not (18%).
Bottom Line
What do we learn from this examination of this large data set arising from approximately 150 laboratories?
- On average, the data precision is about plus or minus 10% when we examine it across laboratories with slightly better values for the macronutrients vs. the micronutrients.
- Collectively, we gain confidence in the capabilities of the agricultural and environmental laboratories participating in the NAPT program. They are capable of excellent precision. For example, the precision for the dry matter method is very good.
- However, some methods have inherent random, indeterminate errors that impact their precision. For example, the analysis for iron is relatively imprecise. This is partially due to known instrument background “noise” that causes fluctuations in the readings. It is also due to the fact that soil has relatively high concentrations of iron and, thus, any dust that finds its way into the digestion of the plant material represents potential slight variations in the analysis.
This data does not give us a measure of any one laboratory, but rather an overview of the laboratory community. Within a laboratory, the precision (and accuracy) can be much tighter than what is shown here. Clients of laboratories can work together for quality assurance as it is a reasonable expectation as part of the quality assurance/quality control relationship.
Examples
So, what does this data mean to us as consumers of laboratory data? Let’s examine a few analytes.
In order to evaluate a laboratory’s accuracy on any one analyte, the value it obtains on a sample is compared with the median of the entire group with plus or minus 2.5 or 4.0 times the MAD. A data point that is more than 4.0 times the MAD above or below the median value is considered to be an extreme outlier (failure for that data point). If it is more than 2.5 times the MAD, but less than 4.0, it is considered a “warning” value. For example, if the median for an analyte was 100 parts per million (ppm) and the MAD was 2 ppm, anything within ± 2.5 MAD would be acceptable. In this case, it would be an acceptable range of 95–105 ppm. Anything outside of ± 4.0 MAD would be a failure, which in this case, would be anything less than 92 ppm or greater than 108 ppm; with anything in between as a warning value.
Understanding Parts Per Million
The term “parts per million,” which is abbreviated as “ppm,” is a measure of concentration. The unit ppm is similar in concept to “percentage” (%). Percentage is parts per hundred, such as 1% is 1 part per 100. A value of 1 ppm is 1 part per 1,000,000. For example, a value of 25 ppm of zinc in plant tissue means that for every million atoms/molecules, 25 of them are zinc. We use percentages to represent relatively larger concentrations, such as the macronutrient concentrations in plants (nitrogen, phosphorus, potassium, sulfur, calcium, and magnesium). We use ppm to express very small concentrations, such as the micronutrients (zinc, iron, manganese, copper, boron, etc.).
We’ll use manganese by the dry ash method as an example as it has a precision score that is about average for the group (note: there are three methods1 used by the NAPT program for digesting plant tissue for analysis, also including nitric acid with either peroxide or perchloric acid). The median value across all of the data evaluated for this exercise was 62 ppm with a MAD of 6 ppm. This means that a laboratory would need to be within 46–77 ppm to be considered passable. Anything outside the range of 36–87 ppm would be beyond the 4 MAD limit. Again, accuracy and precision within a laboratory can and should be much tighter than these values, but this data gives us an understanding of precision across the entire laboratory community.
Another example with an analyte of importance, but with relatively poor precision, is nitrate-N as determined by the Cd reduction method. The median for the data is 205 ppm with a MAD of 29 ppm, giving a range of 134–277 ppm and extreme outliers below 91 and above 319 ppm. This suggests that interpretation across laboratories for this method is somewhat imprecise and needs to be factored in during the interpretation phase. There may be an opportunity to conduct research to improve the interlaboratory precision for this analyte.
In contrast, total nitrogen by the dry combustion method has a median of 2.62% with a MAD of 0.052%, with an acceptable range of 2.49–2.75% and extreme outliers below 2.41 and above 2.83%. This relatively tight range gives added confidence for interpretation of this very important analyte, and once again, shows that laboratories are capable of excellent data analysis if the method is not fraught with inherent random errors. This data would further suggest that researchers should opt to do total N analysis for calibration work over nitrate whenever possible.
Summary
The carpenter’s adage of “measure twice and cut once” is an important one in most things we do. But how precise is the tape measure capable of providing that information? Some yardsticks have wide measurement lines and are somewhat imprecise but still give valuable information in general. Others have very fine lines of demarcation that enable greater precision. In plant tissue analysis, we need to have an understanding of what is realistic with regard to the precision provided by laboratories and not attempt to interpret the results to a finer scale than what is actually achieved.
As a group, the agricultural and environmental laboratories doing plant analysis are capable of great precision. However, some methods, such as dry matter and total nitrogen, have good precision and others, such as iron and nitrate, are relatively less precise due to random, indeterminate errors inherent in the methodology. On average, laboratory data precision is about ± 10%. In general, the macronutrients have more analytical precision than the micronutrients. Plant tissue analysis is a proven, valuable tool to assist in the management of plants and soils. But, its interpretation needs to done with an eye towards actual, measured precision with interlaboratory methods.
Text © . The authors. CC BY-NC-ND 4.0. Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.